
Justification for the Use of Statin in Prevention: An Intervention Trial Evaluating Rosuvastatin (JUPITER): Rationale and Prespecified Analyses What Were the JUPITER Primary Results? Did Early Stopping Impact on the JUPITER Results? Should You Take Statins? Study Says Heart Benefits Outweigh Diabetes Risk The new findings clarify the Type 2 diabetes risks associated with statins, but the debate over who should be taking the cholesterol-lowering drugs is likely to persist. In JUPITER, 75 patients treated with rosuvastatin experienced a nonfatal MI, nonfatal stroke, or hospitalization for unstable angina; underwent revascularization; or died from. 
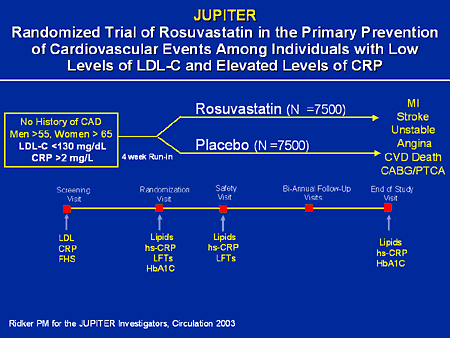
The JUPITER: Rosuvastatin in healthy patients with elevated CRP levels . In patients with average LDL cholesterol levels and elevated CRP, rosuvastatin significantly reduced the risk of a first major cardiovascular event compared to placebo. Rosuvastatin therapy also significantly reduced the risk of all- cause mortality compared to placebo. The study was terminated early and did not meet the prespecified number of primary endpoints needed to be sufficiently powered. Original Date of Publication: November 2. Study Rundown: Statins are commonly used medications in the treating patients with vascular disease and known hyperlipidemia. It had been noted previously that many myocardial infarctions and strokes occur in patients with normal levels of low- density lipoprotein, and in whom, statin therapy would not be warranted based on guidelines at the time. Similarly, C- reactive protein (CRP), a marker of inflammation, has been shown to be a predictor of vascular events. The Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin (JUPITER) sought to determine whether statin therapy could prevent major cardiovascular events in patients with LDL cholesterol levels below treatment thresholds and elevated CRP levels. In summary, the JUPITER demonstrated that treating patients with LDL cholesterol below treatment thresholds but elevated CRP levels with rosuvastatin significantly reduces the rate of first major cardiovascular events as compared to placebo (HR 0. CI 0. 4. 6- 0. 6. Of note, the rates of adverse events were similar in the two groups. Interestingly, this trial was terminated early with a median follow- up less than 2 years. As a result, the trial did not have the prespecified 5. Click to read the study in NEJMIn- Depth . In order to be eligible, patients needed to meet age requirements (. Exclusion criteria included previous or current use lipid- lowering therapy, current use of post- menopausal hormone- replacement therapy, evidence of hepatic dysfunction, elevated creatine kinase level, creatinine > 2. L (1. 76. 8 . Moreover, patients with inflammatory conditions (e. Eligible patients were randomized to treatment with either rosuvastatin 2. Follow- up occurred up to 6. The primary outcome was the occurrence of a first major cardiovascular event (i. Patients were followed for a median of 1. The rate of the primary endpoint was significantly lower in the rosuvastatin group as compared with the placebo group (HR 0. CI 0. 4. 6- 0. 6. This was driven by significant reductions in all components of the primary endpoint, except for hospitalization for unstable angina (HR 0. CI 0. 3. 2- 1. 1. Notably, patients treated with rosuvastatin experienced a significant reduction in all- cause mortality when compared to those treated with placebo (HR 0. CI 0. 6. 7- 0. 9. The rates of adverse events reported were similar for the two groups (p=0. The rates of myopathy (p=0. Image: PD. No works may be reproduced without expressed written consent from 2minutemedicine. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
September 2017
Categories |
 RSS Feed
RSS Feed
